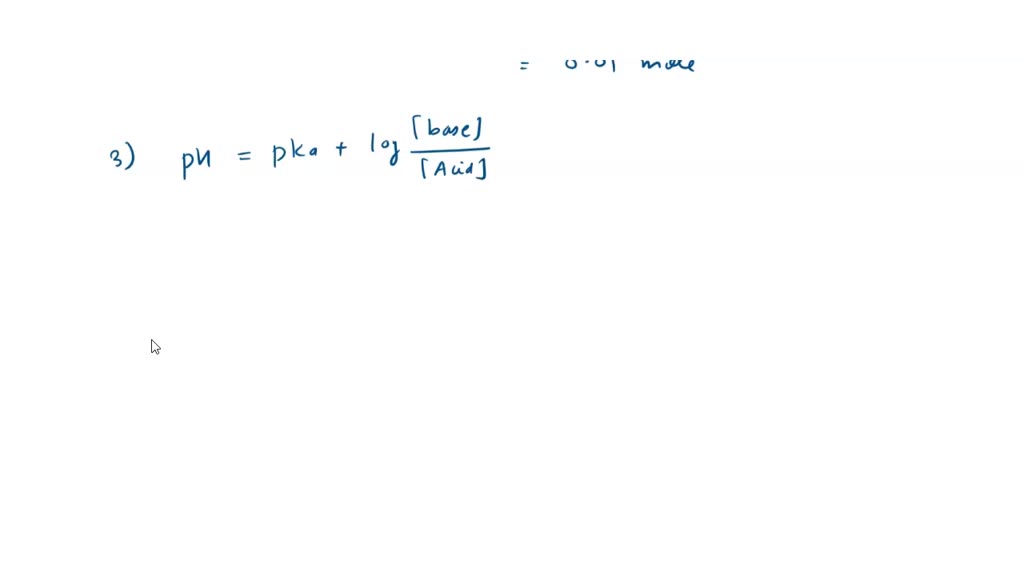
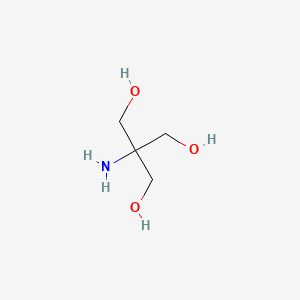
SOLVED: Calculate the masses of tris and tris·HCl needed to make a buffer. Calculate the masses of tris (base, MW: 121.14 g/mol) and tris·HCl (acid, MW: 157.60 g/mol) needed to make 0.10
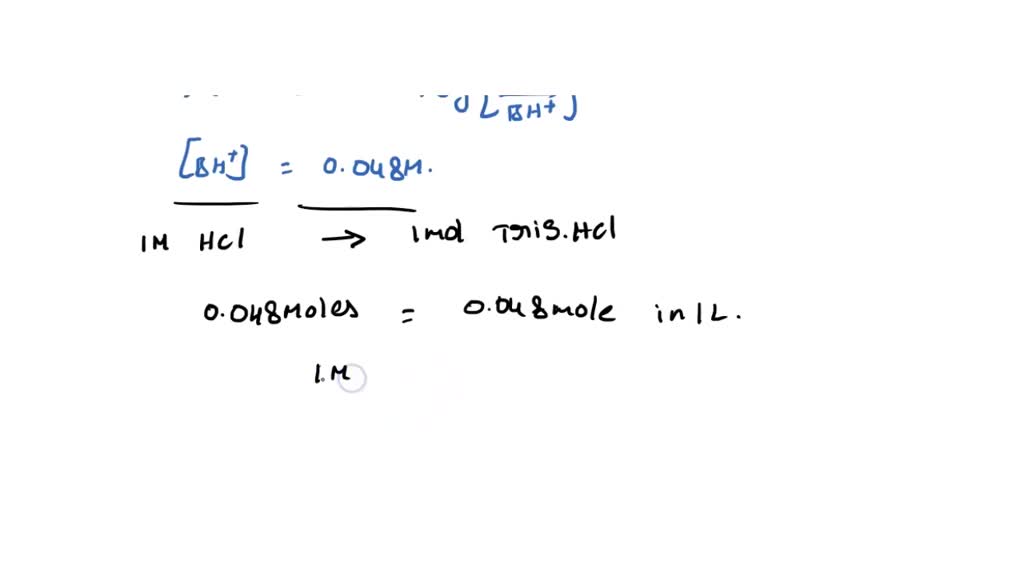
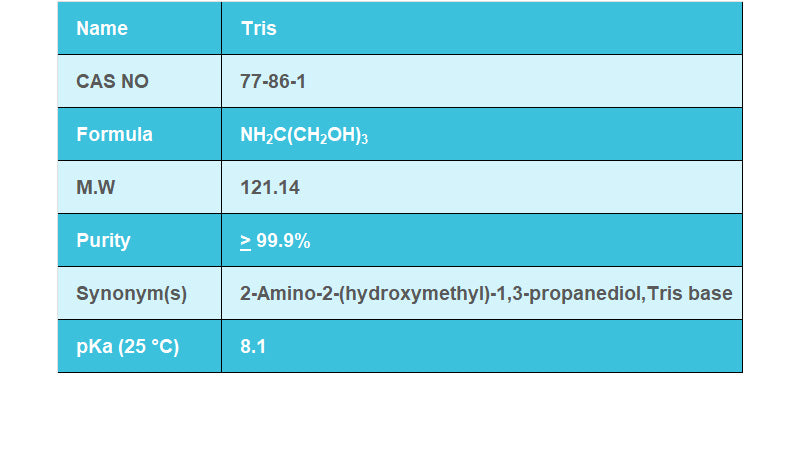
SOLVED: Calculate how you world prepare 100 mL of a 0.1 M TRIS-HCl buffer, pH 7.4 using solid TRIS base (tris-(hydroxymethyl)-aminomethane) (MW 121.1, pKa 8.08). You may find TRIS under several different
![RPI TRIS Base Ultra Pure Powder, 1 Kilogram, Molecular Biology Grade, Buffer Component, [Tris (Hydroxymethyl) Aminomethane]: Amazon.com: Industrial & Scientific RPI TRIS Base Ultra Pure Powder, 1 Kilogram, Molecular Biology Grade, Buffer Component, [Tris (Hydroxymethyl) Aminomethane]: Amazon.com: Industrial & Scientific](https://m.media-amazon.com/images/I/71xgmTB-JtL.jpg)

![T60040-500.0 - Tris Base Ultra Pure [Tris (Hydroxymethyl) Aminomethane], 500 Grams T60040-500.0 - Tris Base Ultra Pure [Tris (Hydroxymethyl) Aminomethane], 500 Grams](https://d2gdaxkudte5p.cloudfront.net/system/images/T60040-500.0_.jpg)
![Tris Base Ultra Pure [Tris (Hydroxymethyl) Aminomethane] Tris Base Ultra Pure [Tris (Hydroxymethyl) Aminomethane]](https://www.spectrum-scientifics.com/v/vspfiles/photos/7124-2.jpg)



![T60040-1000.0 - Tris Base Ultra Pure [Tris (Hydroxymethyl) Aminomethane], 1 Kilogram T60040-1000.0 - Tris Base Ultra Pure [Tris (Hydroxymethyl) Aminomethane], 1 Kilogram](https://d2gdaxkudte5p.cloudfront.net/system/images/T60040-1000.0_.jpg)
![Tris Base [C4H11NO3] Molecular Weight Calculation - Laboratory Notes Tris Base [C4H11NO3] Molecular Weight Calculation - Laboratory Notes](https://www.laboratorynotes.com/wp-content/uploads/2022/11/tris-base-molecular-weight-calculation-300x204.jpg)

![Tris Base (10 kg) [CT01-10KG] - $445.00 : Bioland Scientific, for Your Research Needs Tris Base (10 kg) [CT01-10KG] - $445.00 : Bioland Scientific, for Your Research Needs](https://www.bioland-sci.com/images/Tris%2010kg.jpg)






-500x500.jpg)

![Tris Hydrochloride [C4H11NO3.HCl] Molecular Weight Calculation - Laboratory Notes Tris Hydrochloride [C4H11NO3.HCl] Molecular Weight Calculation - Laboratory Notes](https://www.laboratorynotes.com/wp-content/uploads/2022/11/tris-hydrochloride-molecular-weight-calculation-300x225.jpg)