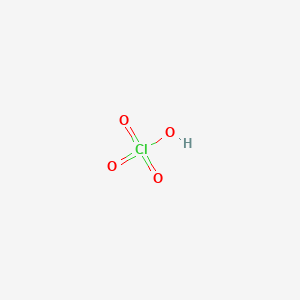
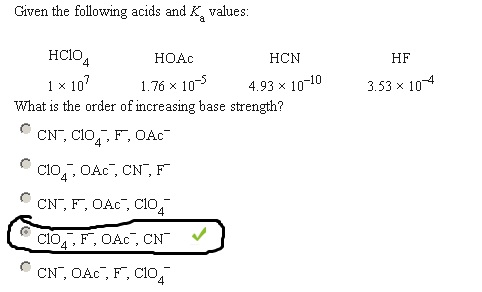
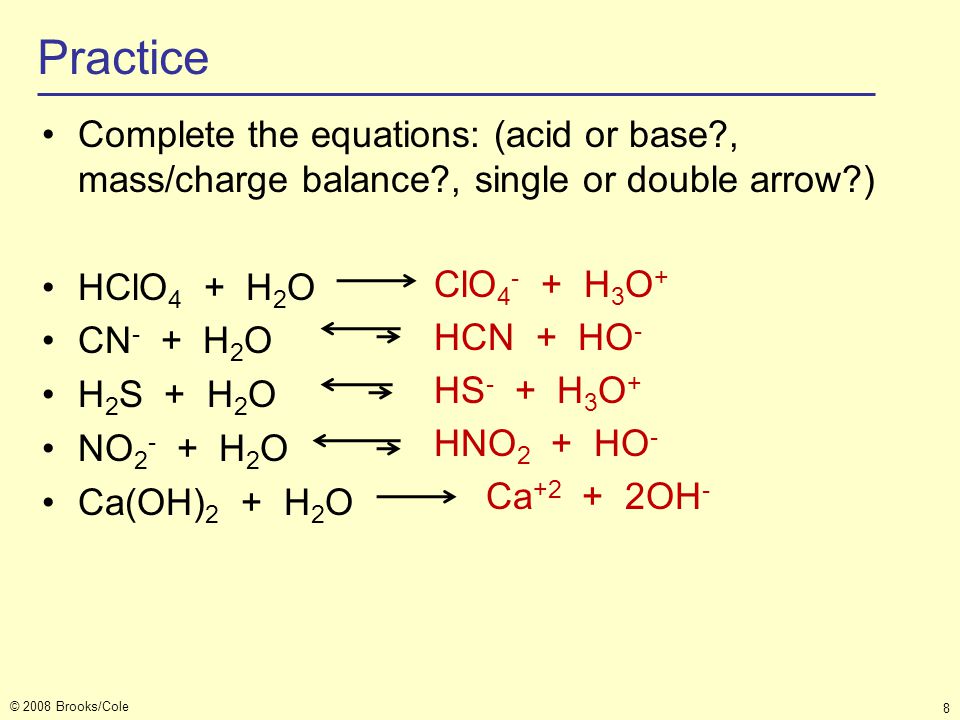
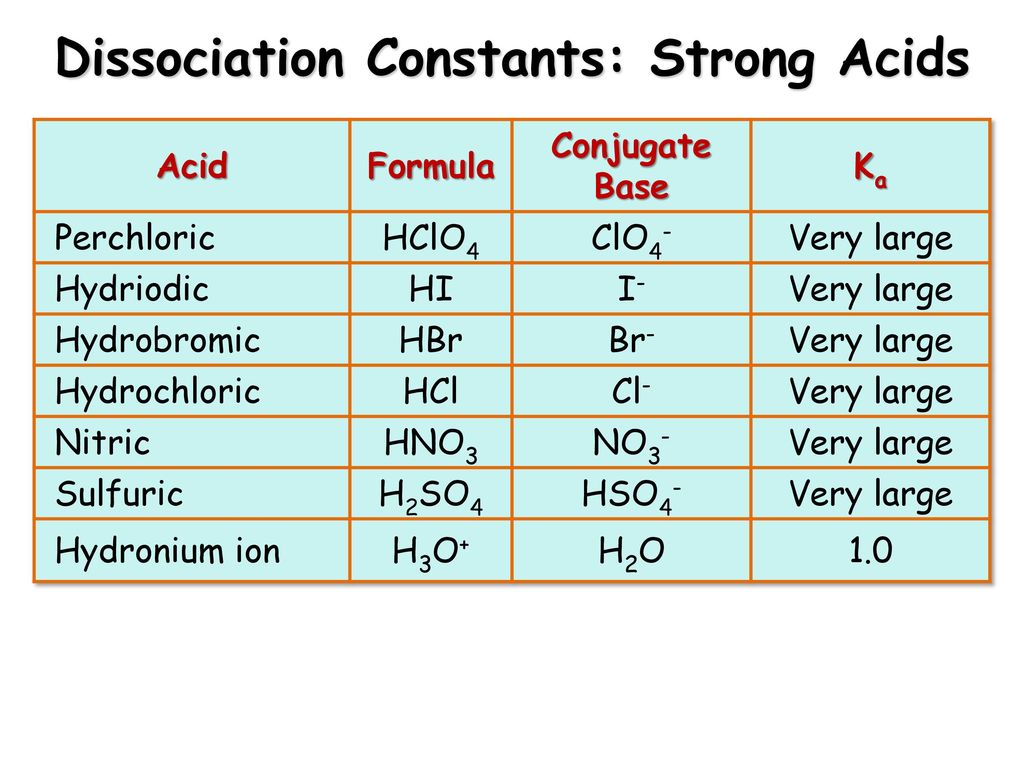
What is meant by the conjugate acid-base pair? Find the conjugate acid//base for the following species: HNO(2), CN^(Θ), HClO(4), F^(Θ), overset(Θ)(O)H, CO(3)^(2-), and S^(2-)

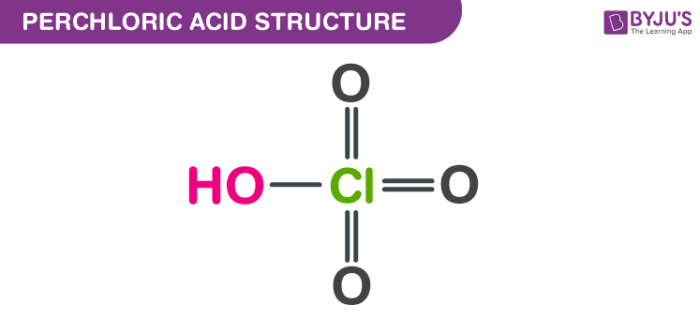
See: Write an equation to show how perchloric acid, HClO4, reacts with water. Include states of matter in - Brainly.com

What is meant by conjugate acid-base pair? Find the conjugate acid/base for the following species.. - YouTube
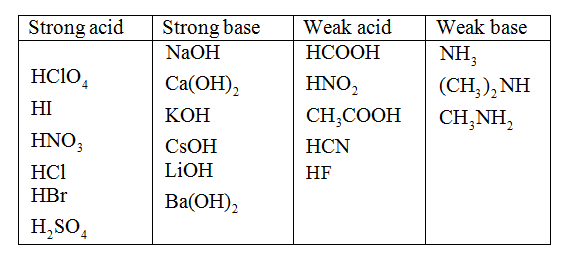
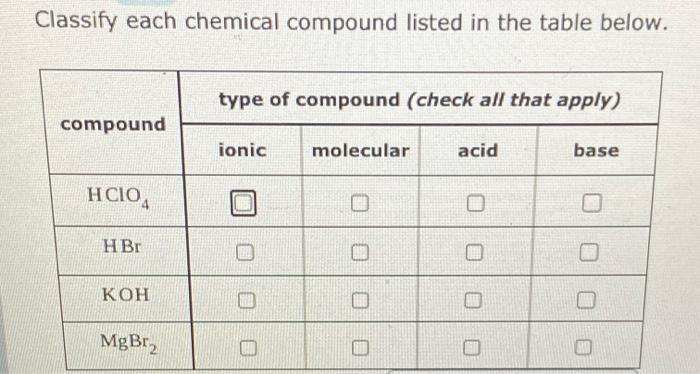
Classify each substance as a strong acid, strong base, weak acid, or weak base - Home Work Help - Learn CBSE Forum
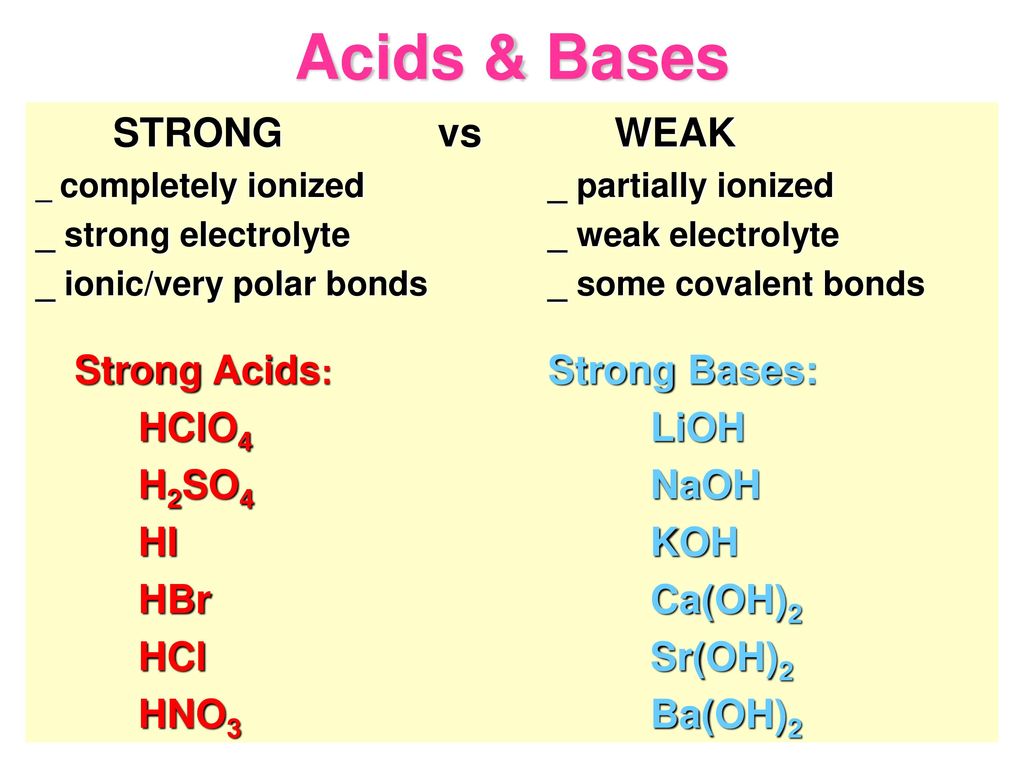
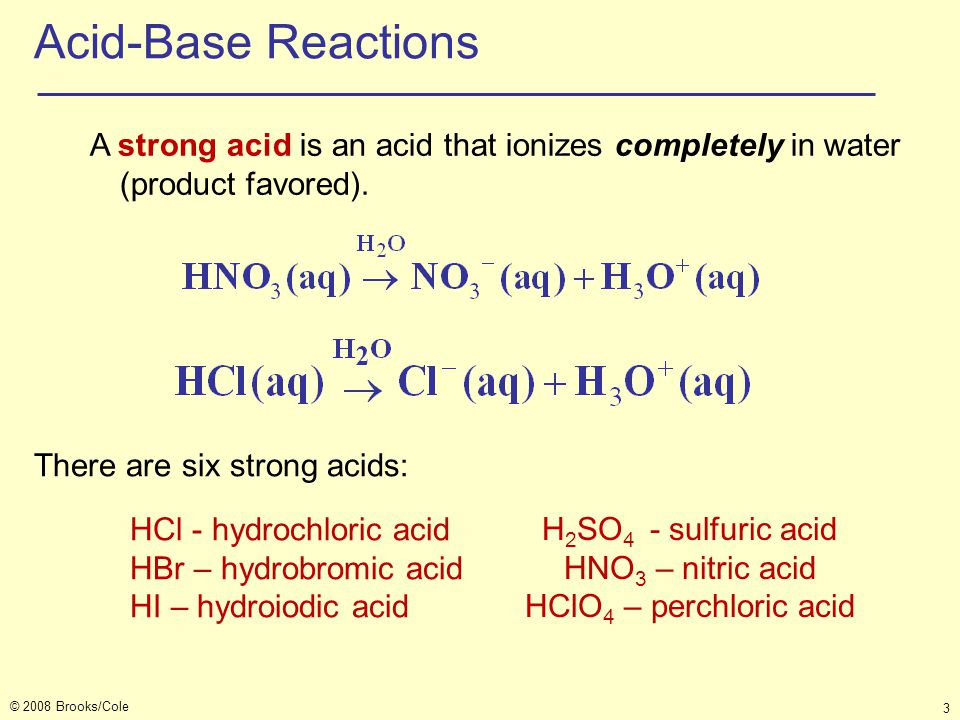
Acids & Bases Acids: acids are sour tasting Arrhenius acid: Any substance that, when dissolved in water, increases the concentration of hydronium. - ppt download